HI Smidge, I'm not sure they would have to do a long term trial of at least a year, indeed, I'm not sure they would have been able to.
I'd be surprised if they were using a new adhesive though. We all know that there exist already sets and CGM sensors which use tried and tested adhesive. I would have approached the manufacturers of these and either used what they have or asked them to develop me something that would last the requisite 14 days using known safe compounds.
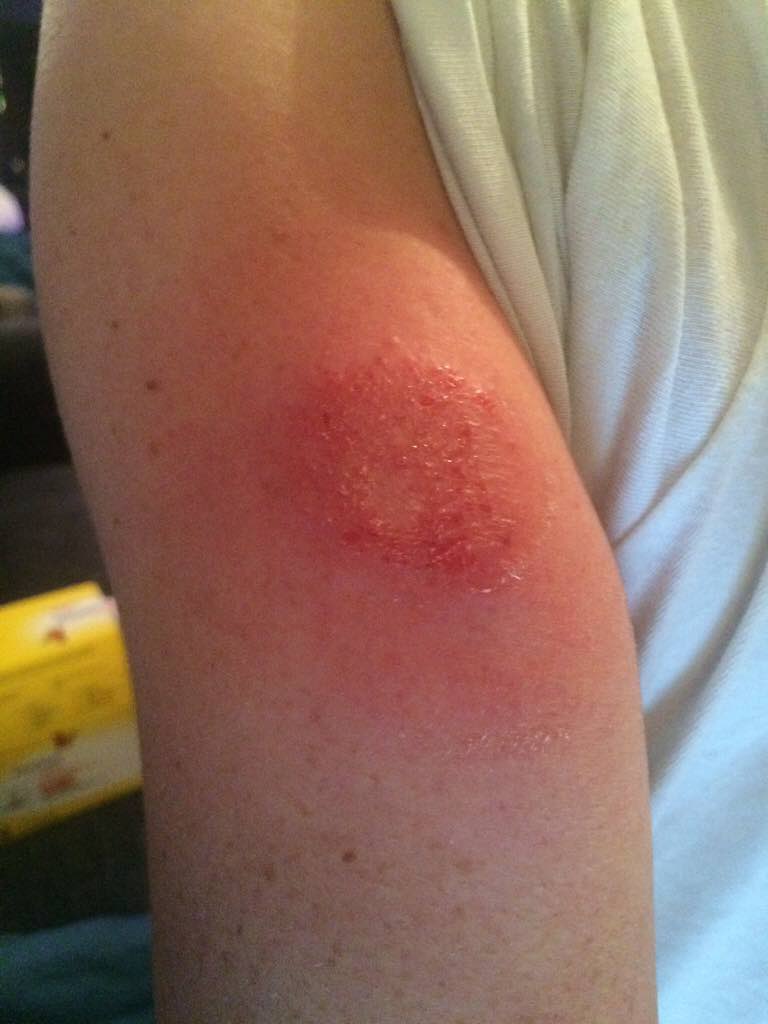
That doesn't mean that there have been extensive tests of the adhesive, and if we go back to the data released by Abbot, 1 out of 30 testers stopped using the system due to skin irritation. If you extend that to (let's guess) 50,000 of us in Europe who have bought in, then it is still more than 1,600 people that could expect to experience skin irritation.
My experience in wearing something like kinesio tape for a long period of time is that your skin does have a reaction to it, simply because it doesn't like having the glue on there, even if there is nothing that shows up at first.
I'd almost certainly class us as the User Test phase of this product release. There is no way that the small sample that they used in the "trials" would have been able to uncover all the potential user issues. I think this is backed up by the level of response that is being received in terms of replacements/exchanges/etc. If you pay a high price, you need to offer this on something so new. I'd hazard a guess that as they iron out the issues, the price will drop and the replacement process will tail off. Think of this as extended R&D if you like!