Johnson and Johnson are set to increase the manufacturing scale of their wearable insulin patch-pen with the aim of launching the product after 2016.
What is the insulin patch-pen?
In 2012, the pharmaceutical company acquired Calibra Medical, who are working on the insulin patch-pen for people with type 1 and type 2 diabetes.
Calibra began developing a three-day patch as an alternative to daily injections of insulin for mealtimes, which was approved in 2010 by the FDA.
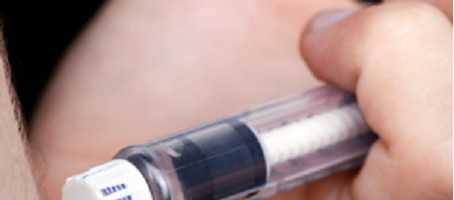
The Finesse insulin patch pen is very small and is worn on the body. It is smaller than an insulin pump and entirely mechanical, with no handheld controller, display screen or batteries.
The patch-pen is designed to deliver bolus insulin doses when two buttons on the device are simultaneously pushed. These can be pushed repeatedly when more insulin is needed than the preset dose.
Basal doses are not delivered by the device, and would need to be administered separately, but the patch-pen would alleviate the pain of daily injections of quick-acting insulin for patients.
The patch-pen has a 200 unit reservoir and can be worn for up to three days, with Johnson and Johnson confident the device will make mealtimes less of a hassle for people with diabetes.
“In terms of the wearable patch pump, that’s probably a couple of years out still, obviously we need to scale up manufacturing complete some additional work on that,” said Dominic Caruso, Chief Financial Offer, during a Q&A session on Johnson and Johnson’s first quarter sales results.
“A few of us were just there recently visiting our diabetes business and all plans are in shape there, they are moving forward with all the manufacturing that needs to be done.”
What's new on the forum? ⭐️
Get our free newsletters
Stay up to date with the latest news, research and breakthroughs.