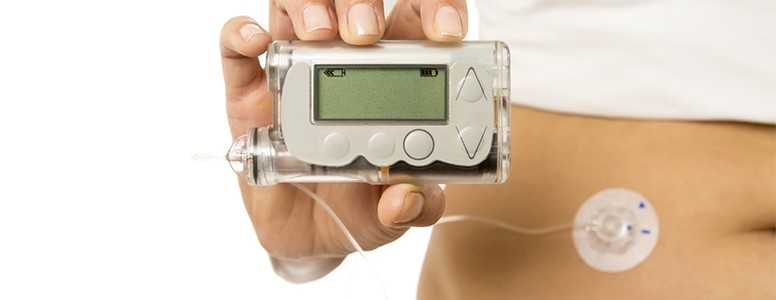
A dual-hormone artificial pancreas using liquid glucagon with the potential to reduce hypoglycemia is undergoing testing in America.
The research could represent a very useful step forwards in the treatment of type 1 diabetes by helping to minimise the risk of severe hypoglycemia.
An artificial pancreas uses continuous glucose monitoring technology to monitor blood sugar levels and insulin pump technology together with computation to adjust the delivery of insulin. This means the device can automatically regulate blood sugar levels and takes away some of the pressure of monitoring blood sugar and estimating insulin doses.
The dual-hormone functionality means that the device is able to release glucagon as well as insulin. Glucagon is useful as it helps to raise blood sugar if levels go too low. Until recently, glucagon was only available in a dry-powder form which is not ideal for use within insulin pump technology.
But now researchers from OHSU (Oregon Health &Science University) School of Medicine are able to use a liquid form of glucagon made by Xeris Pharmaceuticals. The liquid glucagon is stable at room temperature and is suitable for using in an insulin pump.
The study is supported by type 1 diabetes charity, JDRF, and led by Dr Jessica Castle, who is an associate professor of medicine at OHSU School of Medicine. The research will aim to find out if the dual-hormone artificial pancreas together with an exercise detection algorithm is better than a single-hormone artificial pancreas.
The single-hormone artificial pancreas provides insulin but not glucagon and has a low glucose suspend function which switches off insulin delivery if blood sugar levels are about to go too low.
Associate director of research at JDRF USA, Dr Marlon Pragnell said: “JDRF is excited to support OHSU’s research into ready-to-use liquid glucagon. This program has the potential to change the way millions of active individuals with type 1 diabetes monitor and treat their glucose levels.”
Results of the trial are expected to be available in 2019.
What's new on the forum? ⭐️
Get our free newsletters
Stay up to date with the latest news, research and breakthroughs.