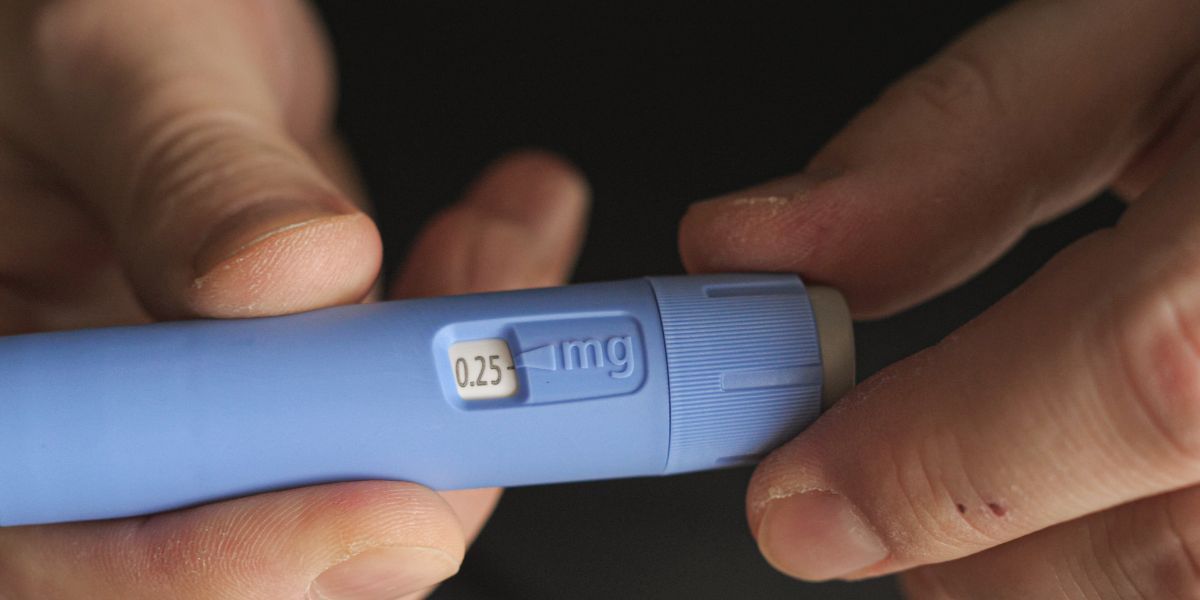
The European drugs regulator is investigating the safety of several weight-loss injections after some people using the jabs have experienced suicidal thoughts.
Safety officers will now review Wegovy, Ozempic and Saxenda – injections used to help people lose weight by suppressing their appetite.
Some people using the weight-loss jabs have also self-harmed whilst using the drug, according to the regulator.
Leaflets on these drugs have already highlighted suicidal thoughts as a potential side effect; however, suicidal behaviour has not been listed for these injections.
- Wegovy: NHS weight loss drug is no magic pill, experts warn
- Wegovy: weight loss drug approved for NHS use
Conducted by the European Medicines Agency’s Pharmacovigilance Risk Assessment Committee (PRAC), the review will examine weight-loss medication that contains semaglutide or liraglutide.
In addition, the PRAC might also assess other medicines, such as glucagon-like peptide-1 (GLP-1) receptor agonists.
A representative from the European Medicines Agency said: “The review is being carried out in the context of a signal procedure raised by the Icelandic Medicines Agency, following three case reports.
“A signal is information on a new or known adverse event that is potentially caused by a medicine and that warrants further investigation.”
They added: “The case reports included two cases of suicidal thoughts – one following the use of Saxenda and one after Ozempic.
“One additional case reported thoughts of self-injury with Saxenda. The European Medicines Agency will communicate further when more information becomes available.”
Over the last six months, weight-loss injections have soared in popularity after some celebrities have used the drugs to rapidly lose weight, such as Kim Kardashian and Elon Musk.
GPs and specialist weight-management clinics in the UK will soon start administering Wegovy and Saxenda to some people.
Common side-effects of the weight-loss injections include stomach ache, headaches, tiredness, diarrhoea, vomiting, constipation and nausea.
Information leaflets on the weight-loss drugs state: “You should pay attention to any mental changes, especially sudden changes in your mood, behaviours, thoughts, or feelings.
“Call your healthcare provider right away if you have any mental changes that are new, worse, or worry you.”
A representative from the manufacturer Novo Nordisk said: “GLP-1 receptor agonists have been used to treat type 2 diabetes for more than 15 years and for treatment of obesity for eight years, including Novo Nordisk products such as semaglutide and liraglutide that have been in the UK market since 2018 and 2009 respectively.
“The safety data collected from large clinical-trial programmes and post-marketing surveillance have not demonstrated a causal association between semaglutide or liraglutide and suicidal and self-harming thoughts.”
They added: “Novo Nordisk is continuously performing surveillance of the data from ongoing clinical trials and real-world use of its products and collaborates closely with the authorities to ensure patient safety and adequate information to healthcare professionals.
“The European Medicines Agency continuously monitors for safety signals and so does Novo Nordisk. Novo Nordisk remains committed to ensuring patient safety.”
Dr Alison Cave, Medicines and Healthcare products Regulatory Agency Chief Safety Officer, said: “As part of our close monitoring, any emerging evidence is routinely considered alongside other sources of information, including suspected adverse drug reactions.
“We will communicate any new advice to healthcare professionals and patients if appropriate. If you are experiencing suicidal thoughts or thoughts of self-harm, please seek immediate medical assistance.”
She added: “We ask everyone to report any suspected side effects using our Yellow Card scheme website.”